Prologue – Eliminating a Surgical Never Event that Should not Exist
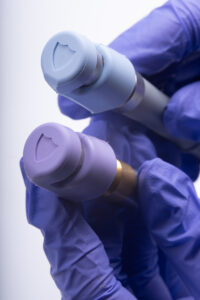
Surgical instruments that emit high-intensity light coupled with human error are a leading cause of intraoperative fires and patient burns. The healthcare system refers to incidents like these as “never events” so they should never happen, right? But, survey results published in the Joint Commission Journal on Quality and Patient Safety share that 1 in 3 nurses have experienced a surgical burn or fire in the past year. This was a red flag for Jackson Medical co-founders James Rains and Kamil Makhnejia.
Rains, Professor of the Practice with the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and GT alumnus Makhnejia began conceptualizing GloShield® to address this safety issue. They brought on to the team Dr. Spencer Kozinn, an Atlanta urologist, to provide clinical expertise to the team.
Together, the nascent Jackson Medical team knew they had a great base concept, but to successfully commercialize the idea, they would need additional outside design and development guidance. That is when they engaged GCMI.
“While completing the Biomedical Innovation and Development Masters Program at Georgia Tech, I worked at GCMI, so when thinking about how to enhance GloShield and build upon the original concept for commercial success, I knew the Phase Zero program at GCMI would be the best option for us,” says Makhnejia.
“The GCMI Phase Zero Program paved a pathway for Jackson Medical to further develop GloShield, an innovative light cable cap that reduces thermal risk in the operating room.”
The Full Pathway – and the importance of quality systems and user needs consideration early
 Ultimately, the Jackson Medical team followed the entirety of the GCMI Commercialization Pathway all the way from Phase Zero through design, development, verification, validation and transfer to production.
Ultimately, the Jackson Medical team followed the entirety of the GCMI Commercialization Pathway all the way from Phase Zero through design, development, verification, validation and transfer to production.
“Based on deep experience and variety in products supported, GCMI knows the requirements for commercialization across the breadth and depth of new medical technologies from the simplest to the most complex. But they know what’s even more important. They know what a technology needs to be within those requirements to actually be successful in the clinic and in the market,” Makhnejia says. “They ensured we designed and built-in usability from Day One. This has had huge benefits. Its straightforward nature and user friendliness are two key reasons GloShield has had solid adoption and sales to date.”
“Understanding user needs must be central to the entire design process. GCMI understands the importance of clearly defined user needs for efficient and comprehensive designs and quality documentation.” – Dr. Emily Blum, MD, Medical Director, GCMI
“Quality management systems – or more explicitly – disregard for quality management systems early in a device’s design and development process can create massive downstream issues the nearer a medical device gets to manufacturing,” Rains says. “Addressing those systems early helped us build a quality-based infrastructure that lasts all the way through launch and market entry making any unanticipated issues much easier and much less costly to address.”

Kamil Makhneija
The Current Status – “Knee deep in sales” and pipeline promise in new national guidelines
According to Makhnejia and Rains, the Jackson Medical team is “knee deep in sales” of the company’s first aforementioned product GloShield. It has been used in more than 150,000 surgeries – with zero burns – nationwide including at Cleveland Clinic, Emory Healthcare, Mayo Clinic, UTSouthwestern Medical Center and Brigham and Women’s Hospital among others.
“GloShield resonates strongly with hospitals and health systems because of the clinical need and device efficacy,” Rains says.
As the American College of Surgeons succinctly put it, “The operating theater has the perfect conditions for fire: ignition sources, oxygen, and fuel.”
As such, associated organizations are taking action.
AORN’s updated Guidelines for Perioperative Practice for 2024 “…encourages adjunct technologies that can reduce the risk of fires. Specifically, they’ve shared that research indicates using a fiberoptic (sic) light cord cover reduces the risk of surgical fires and burns.”
Further, “the Joint Commission recently updated its surgical fire prevention guide for the first time in 20 years. In addition to the actions The Joint Commission requires, they suggest facilities take six additional efforts to prevent surgical fires, [including to] carefully manage electrosurgical devices, light sources and cords, surgical draping, and other risks during a procedure.”
These developments place a bullseye on GloShield as a tool critical to accomplishing the risk of surgical fires and burns per the practice guidelines. GloShield was granted a U.S. patent in April 2023.
“As simply as I can state it, we are currently focused most intently on capitalizing on market opportunities for growth and working with GCMI on two early stage new products complementary to GloShield” Rains says.
4 Things Medtech Innovators Can Learn from Jackson Medical’s Journey Thus Far
- If your technology requires evaluation and approval or recommendation by a hospital’s value analysis committee (VAC), find your champion be it a surgeon, other clinician including nurses, a clinical workflow or patient safety expert for example. They will understand your “why.” Then give them everything they need to educate others and advocate for you in a single source VAC “packet.”
- After a fundraising round, stay hyper-focused on what’s been working well. It’s good to experiment, but do not get distracted. Continue to capitalize on momentum and do more of what’s working with scale. Be smart with the funds and be lean. Being lean has been a critical component to success.
- Selling a product yourself to start teaches you about the buyers’ cycles, about what moves people, about improving your product marketing and ‘positioning’ and giving you confidence in your product and in yourself. It also enables you to know, traverse and eventually avoid pitfalls while keeping you lean as well.
- When it’s time to scale sales, be very selective about partners. It is possible to de-risk sales like investors need de-risked technologies. Know what’s driving sales or distribution partners and how exactly your product or technology fits in their portfolio. Are their call points – the people they are contacting regularly at hospitals – the same as the ones that would be buying decision makers for your device or not? If not, your technology will be a deviation for them, requiring new relationship development and likely selling on aspects or value propositions with which they are not familiar. Not an ideal partner prospect. However, if they have relatively limited numbers of SKUs in their portfolio or catalog, they may well have room to give your product more attention than others. On the other hand, if a distributor has 10,000 products to sell, they will need to be highly incentivized to sell your product. Does it stand out from the rest of their portfolio? If not, they will not represent it the way you want to.
Why GCMI? – Just any guide won’t do for Mt. Everest

James Rains
“Developing and launching new medical technologies including relatively simple devices like GloShield is like climbing Mt. Everest,” Rains says. “View GCMI like the guide you need for such a serious endeavor. They work the strategy, the navigation, the right partners and if you find yourself in a bad spot, they’ll be there with the oxygen you need to get to the summit. And if you find yourself on a seriously challenging path, you won’t know which way to go or just the waypoint you’re looking for. Go talk to them, at minimum, they can serve as a sounding board with serious expertise from beginning all the way through.”
“We can’t get where we want to go, as efficiently and effectively as we need to get there without GCMI,” Makhnejia adds. “And because our operations are based at GCMI’s offices and labs we have the luxury of leaning on their team for guidance or answers to questions of all kinds, manufacturing, quality control, legal or regulatory, almost instantaneously as they pop up.
“The easy access to clinical expertise and tools like state of the art 3D printers for customer demos or promotional leave behinds are very real benefits one might not consider until one has to go searching for them externally.”
Get in touch with GCMI
GCMI is fully committed to our customers’ success and welcomes you to contact us at any point in a technology’s pathway from the ‘back of the napkin’ to the bench, manufacturing, bedside and beyond. Whether you’re an individual innovator, startup or health system with an internal innovation program, initiative or ecosystem, get in touch. It’s never too early.
